What Alumina Is and How It Is Defined in Engineering Contexts
Alumina, also known as aluminum oxide, is an inorganic ceramic compound with the chemical formula Al₂O₃. In engineering contexts, it is classified as an oxide technical ceramic rather than a structural metal or polymer.

Alumina is selected in engineering systems where dimensional stability, wear resistance, electrical insulation, and thermal reliability are required simultaneously. It is not chosen for ductility or impact tolerance, but for predictable performance under heat, load, and chemically aggressive conditions.
Naturally, alumina occurs as corundum. When alloyed with trace elements, corundum forms ruby or sapphire, demonstrating that hardness and crystal stability originate from the same base structure used in technical ceramics.
In industrial use, alumina is supplied as a white or near-colorless crystalline powder and then processed into dense ceramic components. Its insolubility in water and most solvents directly supports its use in chemically stable assemblies.
Core Properties That Define Alumina Performance
Alumina’s engineering value does not rely on a single extreme property. It comes from a stable combination of hardness, temperature resistance, electrical insulation, and chemical stability that defines a clear operating envelope.
Alumina exhibits a Mohs hardness of approximately 9 and a Vickers hardness typically between 15 and 20 GPa. This level of hardness explains its widespread use in sliding, abrasive, and erosive environments such as seals, nozzles, bearings, and wear liners. The same characteristic also limits its suitability for impact-loaded structures, where sudden stress can cause brittle fracture.

Typical flexural strength ranges from 300 to 600 MPa, while compressive strength often exceeds 2000 MPa. Alumina performs reliably under compressive and steady mechanical loads. Fracture toughness remains relatively low at 3.5 to 4.5 MPa·m¹ᐟ², which requires designs that minimize stress concentration and avoid shock-dominated failure modes.
With a melting point near 2072°C and continuous operating temperatures commonly reaching 1500 to 1750°C, alumina replaces metals in furnace hardware, insulation supports, and high-temperature fixtures. Its thermal shock resistance is generally limited to ΔT values around 200 to 220°C, which constrains its use in applications involving rapid or repeated temperature swings.
Alumina provides excellent electrical insulation, with volume resistivity above 10¹⁴ Ω·cm and dielectric strength typically between 10 and 16 kV/mm. This combination makes it suitable for high-voltage and high-temperature electrical systems where polymers or metals would fail.
Chemical inertness further defines alumina’s role. It resists strong acids and alkalis at elevated temperatures and remains stable in most industrial environments. Its biological inertness also supports long-term use in medical implants where material interaction with tissue must remain minimal.
Thermal conductivity usually ranges from 20 to 45 W/m·K. Alumina conducts heat more effectively than polymers but less than materials such as aluminum nitride. This balance supports moderate heat dissipation while maintaining electrical insulation, rather than maximizing thermal transfer alone.
Alumina Types and Purity Grades Used in Industry
Alumina Classification by Purity Level
Ninety-six percent alumina is commonly selected when cost efficiency is important and operating conditions remain moderate. It is widely used for general insulation components and wear parts.
Alumina grades in the 99.5 to 99.7 percent range are chosen for more demanding environments. Higher purity improves corrosion resistance, thermal stability, and wear consistency over long service periods.
Ultra-high purity alumina, typically 99.95 to 99.99 percent, is used when electrical reliability, chemical cleanliness, or biocompatibility cannot be compromised. These grades are standard in semiconductor processing and biomedical applications.
Alumina Classification by Crystal Structure
Alpha alumina, or α-Al₂O₃, is the most stable phase and provides the highest mechanical strength and thermal resistance. It is the preferred structure for load-bearing and wear-critical ceramic components.
Gamma alumina, or γ-Al₂O₃, features a high specific surface area and is selected for catalysis and adsorption rather than structural use.
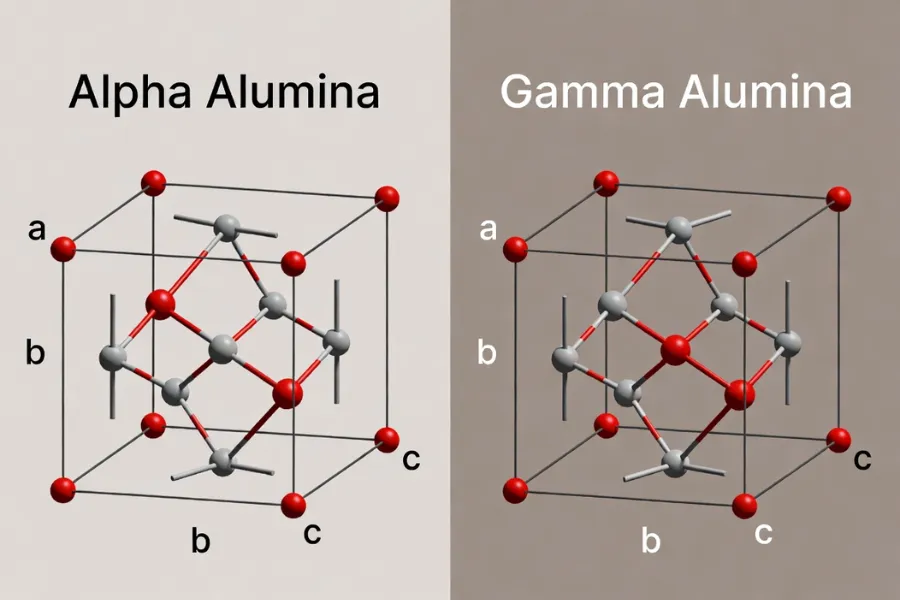
Other transition phases such as theta, delta, and kappa alumina appear during thermal transformation but are rarely used in final engineering components due to reduced stability.
How Alumina Is Produced, The Bayer Process Explained
Most industrial alumina is produced from bauxite using the Bayer process, which enables large-scale output with controlled purity.
Crushed bauxite reacts with concentrated sodium hydroxide at elevated temperature and pressure to form sodium aluminate. Insoluble residues are removed through filtration, leaving a purified solution.
Aluminum hydroxide is precipitated by controlled cooling and seeding. High-temperature calcination removes chemically bound water and converts the hydroxide into alumina powder.
Calcination conditions determine the final product. Activated alumina remains porous and is used for adsorption and catalysis. Smelter-grade alumina feeds aluminum electrolysis. Fully calcined alumina is used for ceramic and refractory applications.
Where Alumina Is Used in Industrial and Technical Applications
Alumina is used in electronics and semiconductor manufacturing for substrates, packages, and insulators where electrical isolation must be maintained under thermal load.
In industrial ceramics and mechanical systems, alumina components such as seals, bearings, and nozzles are selected when abrasion resistance and dimensional stability matter more than impact tolerance.
Refractory systems rely on alumina for furnace linings, casting systems, and high-temperature fixtures exposed to heat and corrosive atmospheres.
Medical and biotechnology applications use alumina in joint replacements, dental components, and surgical tools due to its biocompatibility and low wear debris generation.
In chemical and environmental processing, gamma alumina supports catalysts and adsorbents, while dense alumina protects equipment from corrosive fluids and gases.
Aerospace and defense applications use alumina in thermal protection systems and armor where hardness and heat resistance outweigh toughness requirements.
Automotive systems employ alumina in sensors and exhaust components exposed to high temperature and chemical attack.
Abrasives and surface finishing processes continue to rely on alumina for grinding, polishing, and controlled material removal.
Common Engineering Questions About Alumina
Is alumina safe to handle
Solid alumina is generally non-toxic and safe under normal industrial handling. It does not react with skin or most chemicals. However, inhalation of fine alumina dust should be avoided, as airborne particles may irritate the respiratory system. Standard dust control and ventilation practices are recommended during processing and machining.
Why does alumina cost more than common ceramics
Alumina production involves energy-intensive refining, high-temperature calcination, and controlled sintering. Costs increase further for high-purity grades and tight-tolerance components that require diamond grinding. These factors raise manufacturing expense but also deliver consistent performance in high-temperature, high-wear, and electrically demanding applications.
How does alumina differ from aluminum metal
Aluminum is a ductile, electrically conductive metal used for structural and conductive applications. Alumina is aluminum oxide, a hard, brittle ceramic that provides electrical insulation, wear resistance, and thermal stability. The two materials serve entirely different engineering roles despite sharing the same base element.
Can alumina be recycled
Alumina is chemically stable and environmentally inert. Manufacturing scrap and certain post-use ceramic components can be recycled or reused in secondary applications. While fully sintered alumina cannot be remelted like metals, material recovery is possible through crushing and reprocessing in suitable industrial workflows.
What is alumina’s main limitation
The primary limitation of alumina is brittleness. While it performs well under compressive and steady loads, it is unsuitable for impact-dominated or shock-prone applications. Its high hardness also complicates machining, often requiring diamond tools, which increases cost for complex geometries.
How should alumina purity be selected
Purity selection depends on operating conditions. Ninety-six percent alumina suits general wear and insulation. Purities above 99.5 percent are chosen for high-temperature, corrosive, or high-voltage environments. Ultra-high purity grades are required for semiconductor and biomedical applications where contamination and electrical reliability are critical.
Future Development Trends and Engineering Summary
Future development of alumina focuses on higher purity levels, improved forming techniques, and composite systems such as zirconia-toughened alumina to enhance fracture resistance. Applications in semiconductor manufacturing, energy systems, and biomedical engineering continue to expand.
From an engineering perspective, alumina is selected when hardness, temperature stability, electrical insulation, and chemical resistance must coexist in a single material. It is avoided where impact tolerance or extreme thermal cycling dominates design constraints.
Suppliers such as C-CERAMIC support these requirements by providing alumina ceramics across a wide range of purities and component geometries, enabling engineers to align material selection with real operating conditions rather than nominal specifications.






